Home > Immunology Assays > T Cell Activation Assays
Assays for measuring T cell activation, killing, proliferation, signalling and synapse formationCelentyx offers high-throughput plate-based and high-content assays of T cell activation, including on purified T cell subsets. We use multiple different readouts and conduct studies on human primary cells from both healthy volunteers and patients with underlying disease.
Assays include: 1. T cell activation 2. T cell killing 3. Mixed Lymphocyte Reaction (MLR) 4. Antigen-dependent T cell activation 5. Immune synapse localisation 6. T cell polarisation Assays may be suitable for evaluating modulators of BiTEs, CD3, CD27, CD28, CD40, CD40L, CD70, CD80, CD86, CTLA-4, JAK1, JAK2, JAK3, ICOS, ICOSL, IL-2R, IL-9R, IL-21R, OX40, OX40L, PD-1, PDL-1, TIGIT, TIM-3, STAT1, STAT3, STAT5 and others |
Further Immunology Assays
Antigen-specific Assays B Cells Fibroblasts Haemolysis Testing Human Microglia Macrophages/Monocytes Neutrophils/Granulocytes NK Cells Phagocytosis Assays Regulatory T cells Spheroid Killing Assays Suppression Assays T Cell Exhaustion Assays TIL and dissociated tumour cell assays Tumour Cell Killing Assays |
1. T cell activation |
2. T cell killing |
|
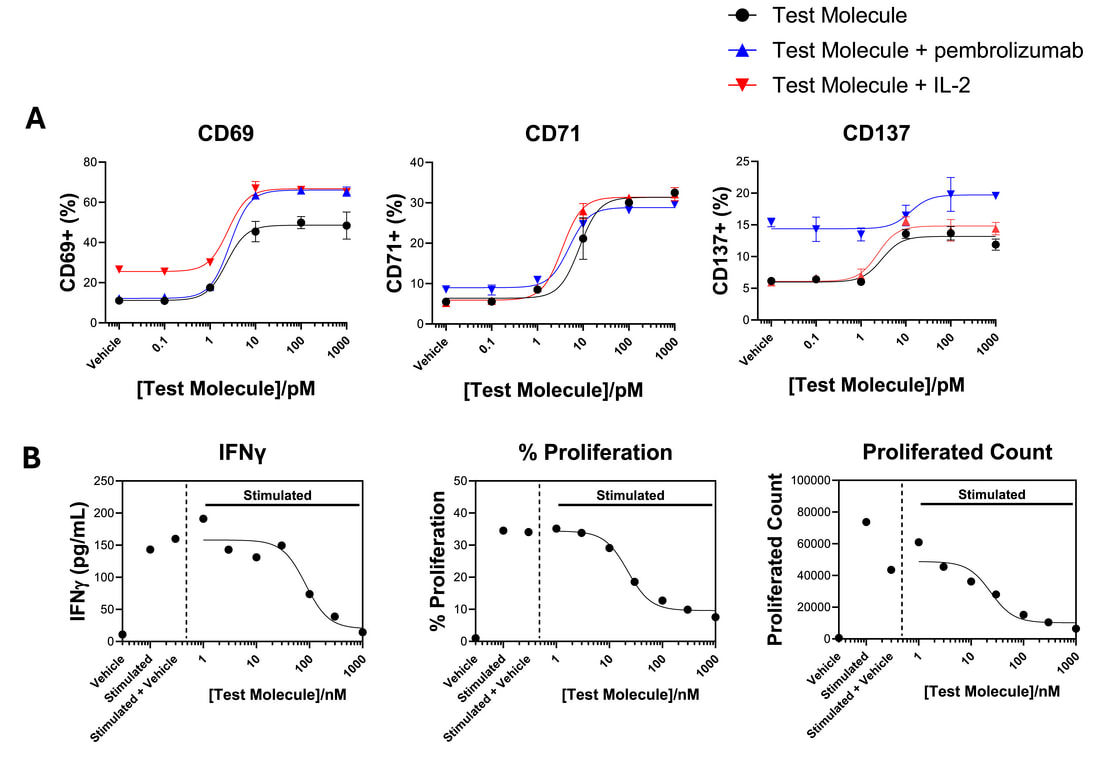
T cells can be activated with a range of stimuli including antigens presented by dendritic cells, recombinant stimuli that engage CD3 and CD28, and tumour cells amongst others. A Shows activation of CD4 T cells in the presence of tumour cells by a test molecule as reported by increase in activation markers quantified by flow cytometry. The presence of IL-2 or pembrolizumab modulate this response. B Shows a JAK inhibitor reducing a CD8 T cell cytokine (IFNγ) and proliferation in a dendritic cell driven response.
|
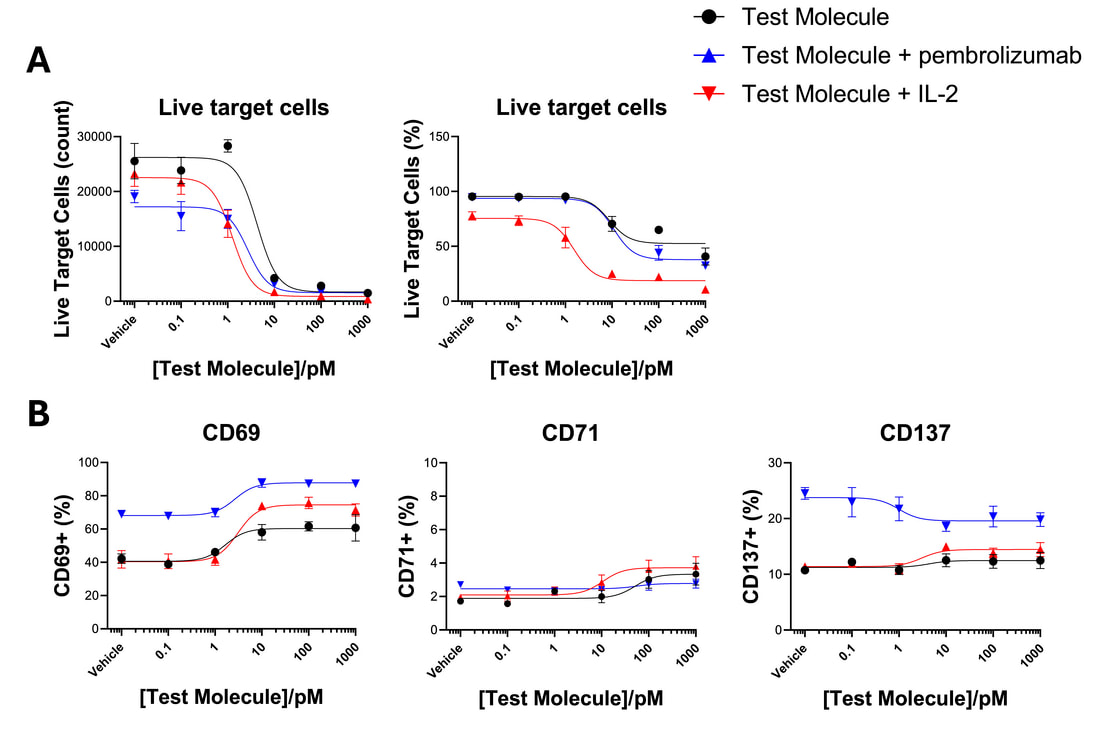
Cytotoxic T cells, such as CD8 T cells can be used as effector cells, and their ability to kill target cells, such as tumour cells can be monitored by flow cytometry. This allows quantification of both target cell killing (A) and immune cell activation (B). Killing assays can also be run by imaging (see our dedicated tumour cell killing page). A range of cytotoxic T cells can be used, including CD8 T cells, γδ T cells and mucosal-invariant T cells (‘MAIT’ cells). The example below shows killing of target cells by CD8 T cells evoked by a bispecific engager (BiTE) and modulation by IL-2 and pembrolizumab, while also monitoring T cell activation.
|
3. Mixed-lymphocyte reaction (MLR)
|
A dendritic cell-driven ‘mixed lymphocyte reaction’ or ‘MLR’ enables the impact of test molecules targeting T cells or dendritic cells to be evaluated. Multiple aspects of the T cell response can be investigated including activation marker expression, proliferation, differentiation and cytokine production. The example on the right shows the impact of two reference immunosuppressive agents on CD4 and CD8 T cell activation markers, and on cytokine (TNFα and IFNγ) production.
|
4. Antigen-dependent T cell activation
|
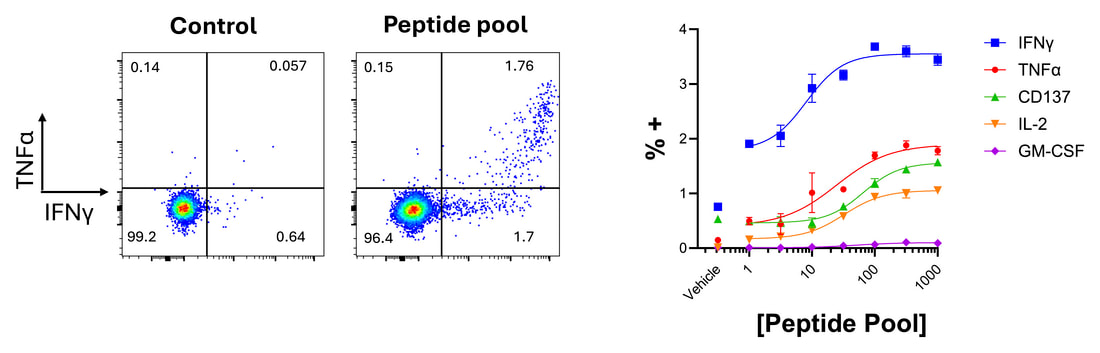
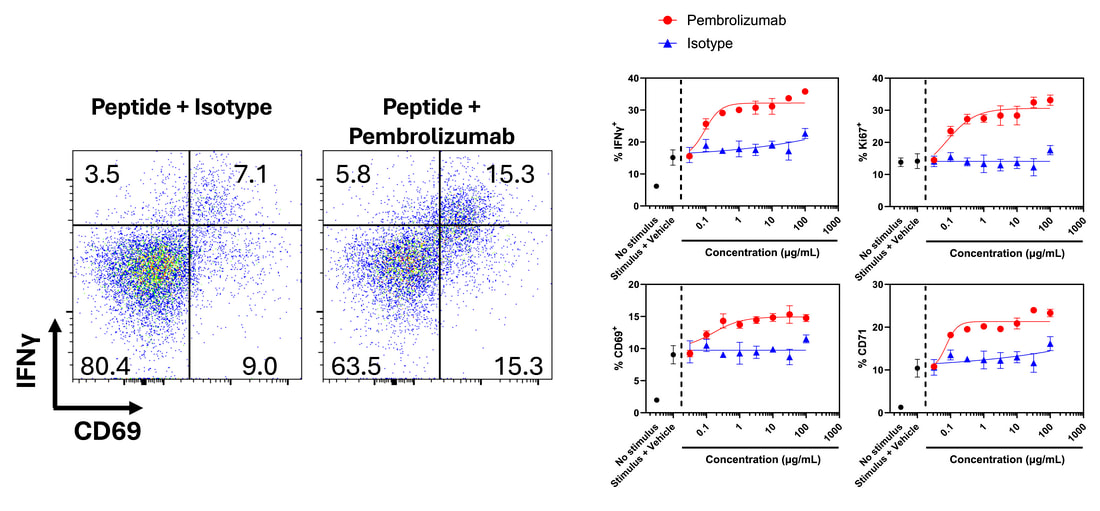
Peptides and protein antigens can be utilised to evoke antigen-specific responses of T cells (also see our dedicated antigen-specific responses page). When combined with phenotypic/exhaustion markers and intracellular cytokine labelling, this approach can be used to understand what cell subsets are responding, and to investigate modulation of the response by test agents.
|
5. Immune synapse localisationA number of assay systems can be utilised to investigate recruitment of receptors to the immune synapse, and disruption/modulation of this process by therapeutic agents. Also see our dedicated imaging page.
|
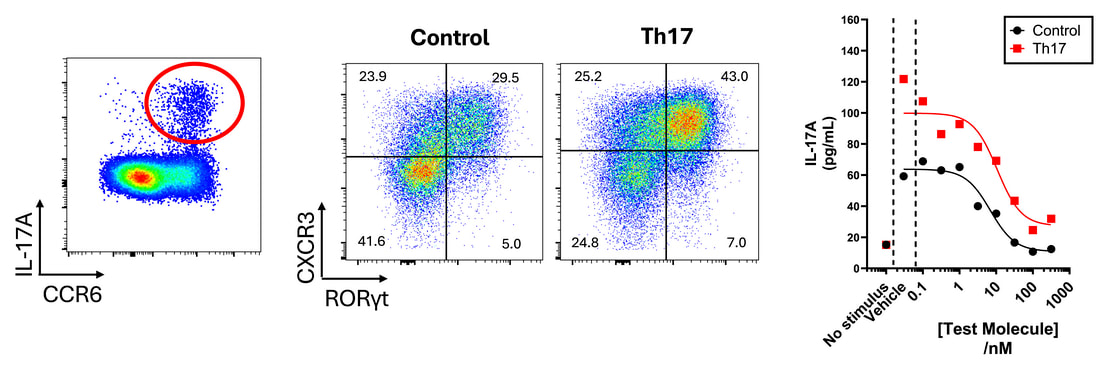
6. T cell polarisationBy culturing T cells in defined cocktails of cytokines, polarisation of T cells into key subsets including Th1, Th2, Th17 and Tregs can be achieved (also see our dedicated regulatory T cell page). This polarisation can be monitored by flow cytometry following transcription factor labelling, and the impact of test compounds assessed both on polarisation and on subsequent functional responses. The example below highlights IL-17 production (by intracellular cytokine staining) and Th17 polarisation (confirmed by increased RORγt expression), and the inhibition of IL-17 production by a test molecule.
|